Do you and ILIAD have Chemistry?
Many may find it surprising, but optimization plays many valuable roles in the field of chemistry. One such application is in predicting equilibrium concentrations of chemical mixtures. At a given temperature and pressure, a mixture moves towards equilibrium by lowering its Gibbs free energy which occurs through a fluctuation in the concentrations of its constituent species. Gibbs free energy can be expressed mathematically as:

Or alternatively:
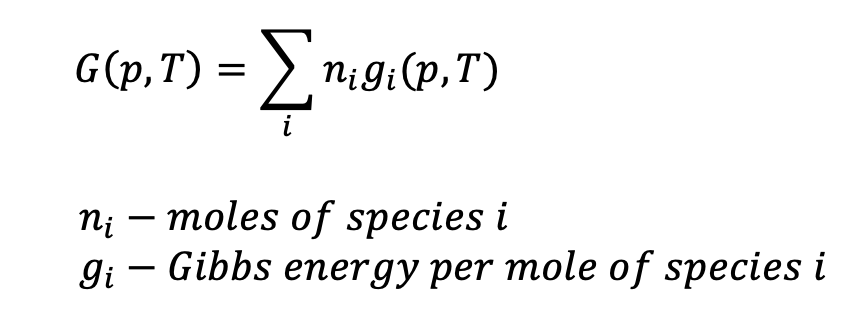
The equilibrium concentrations of each chemical species can be found by solving a minimization problem for Gibbs energy without ever having to consider all the possible intermediate reactions!
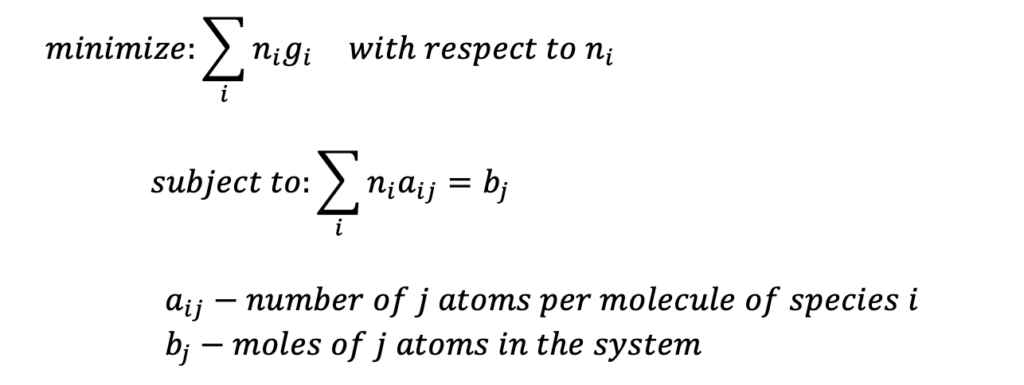
In plain English, this means: ‘find the amount of each kind of molecule that minimizes the total Gibbs energy while still preserving the number of each type of atom involved in the mixture.’ Thermodynamic properties such as enthalpy, and entropy are complex functions of temperature and pressure (interested readers see Nasa Polynomials) so accurately predicting equilibrium concentrations manually is largely infeasible for even moderately sized mixtures, but not so for ILIAD!
As an example, imagine a 10𝑚 storage tank with a gas mixture initially with 4 moles each of 𝑂2, 𝐻2, 𝐻2𝑂, 𝐶𝐻4, 𝐶𝑂, and 𝐶𝑂2 held at a steady 600K. Despite the complexity, ILIAD’s optimizer takes only a few seconds to compute that the equilibrium concentrations are 200ppb, 250ppb, 38,000ppm, 19,000ppm, 1,500ppb, and 39,000ppm respectively. Using the ideal gas relation with the resultant mixture, we also find that there is a small pressure increase of roughly 60Pa, a useful piece of knowledge if we are choosing or working with a pressure vessel.
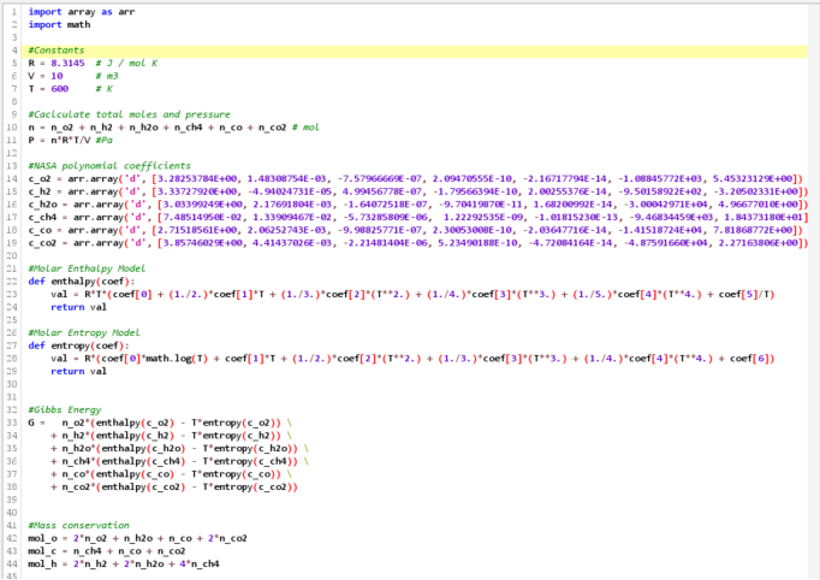
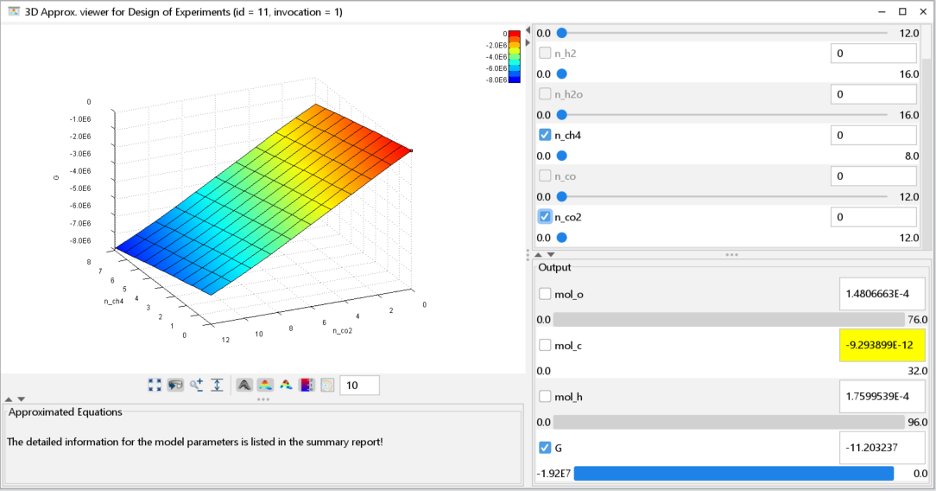
ILIAD’s response surface modeling and visualization tools help us better understand the effect each gas species has on the total Gibbs energy of the mixture.
n this case we can see that 𝐶𝑂2 has a much steeper slope than 𝐶𝐻4 or 𝐶𝑂 do at this temperature, so at equilibrium, carbon will tend to form much more 𝐶𝑂2 rather than the other carbon species if possible.
Check back in weekly for more tips, tricks, and interesting case studies using ILIAD and Design Studio with GENESIS!

